From Drive Medical
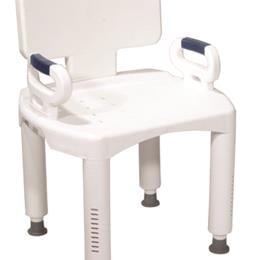
The Drive Medical Shower Chair with Back and Arms is designed for people with mobility limitations and comfortably accommodates users of all sizes. View more
Bath Bench Premium Series
The Drive Medical Shower Chair with Back and Arms is a premium bathroom product that can help provide additional safety and stability while you're showering. This high-quality shower seat helps users maintain their independence, ensuring that they have the support they need when entering, exiting, or sitting in the tub. The Premium Series Shower Chair is designed for people with mobility limitations and comfortably accommodates users of all sizes.
This shower seat is both lightweight and stable, sporting large legs with non-slip padding on the feet. The height of the chair can be easily adjusted without the use of tools, and the back, legs, and arms can be quickly and conveniently removed. This makes the Drive Premium Series Shower Chair one of the most portable and easy-to-store shower seats on the market.
Durable, versatile, comfortable – the Premium Series Shower Chair from Drive is everything you or a loved one needs in a shower safety seat.
Product Details
| Prod Spec | Prod Spec Value |
|---|---|
| Manufacturer SKU | 1199 |
| Length | 22.2" |
| Width | 21.75" |
| Height | 17.9" |
| Weight | 10 lbs. |
| Weight Capacity | 350 lbs. |
- Tool free height adjustment (1/2” increments)
- Comfortable seat accommodates users of all shapes and sizes
- Back, legs, and arms can be removed without the use of tools
- Lightweight design
- Large legs with non-slip padding on feet